Digital-first electronic
outcomes for any trial.

Catchtrial® Apps+ replace paper documents and fragmented tools with a secure, intuitive digital experience. Patients and clinicians capture outcomes from anywhere, on any device — reducing site burden while improving data quality and protocol adherence.

Designed For Patients.
Built For Real Insights.
Catchtrial® enables secure patient onboarding and end-to-end data oversight through a privacy-first architecture designed to meet global regulatory and inspection-readiness requirements.

Capture Outcomes in Real Time
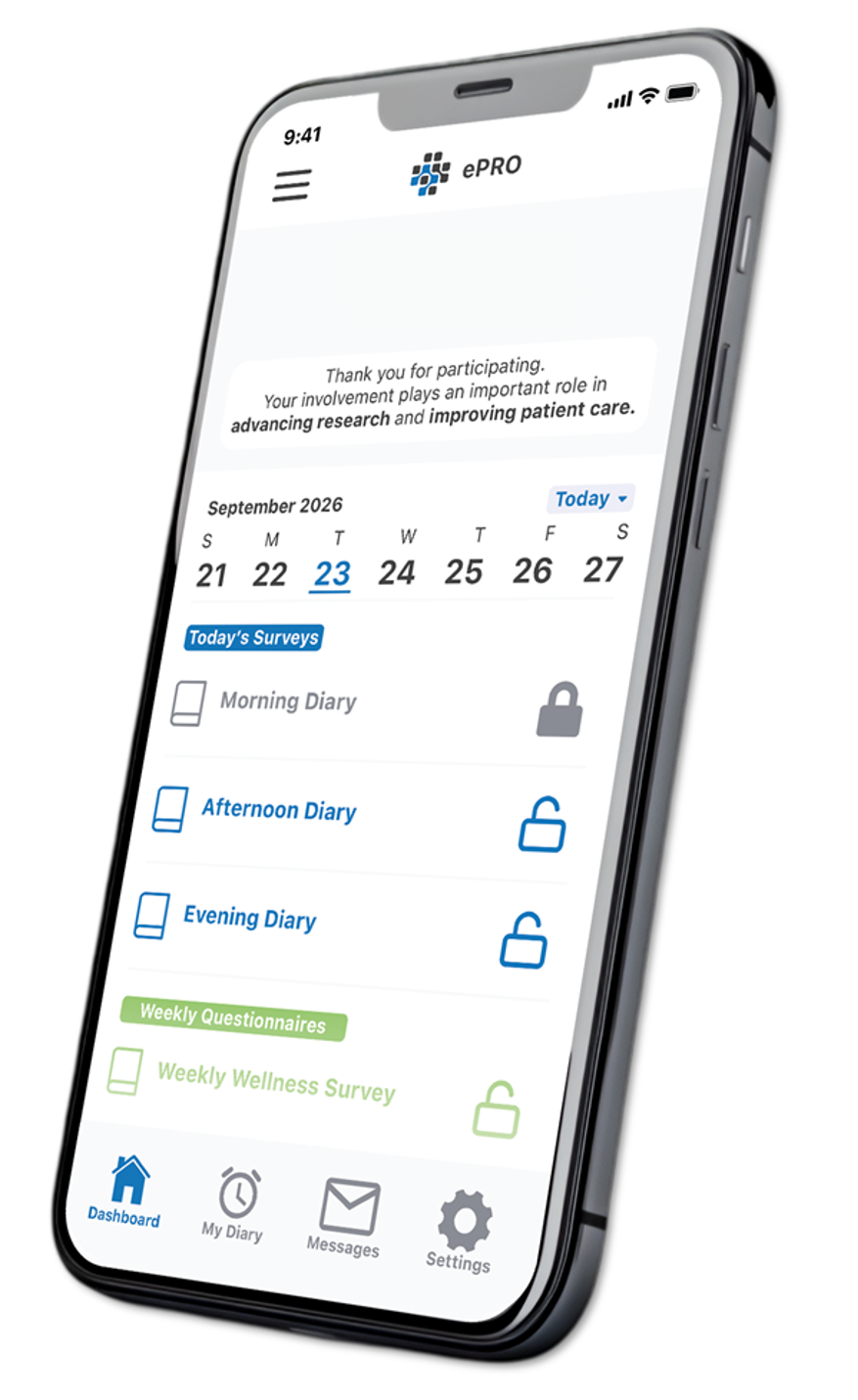
Catchtrial® ePRO delivers health surveys directly to patients on any device. Symptoms, diaries, and quality-of-life data are captured in real time and flow straight into your EDC — no manual re-entry, no delays.

Design Surveys Without Code
The built-in ePRO Forms Builder lets study teams create and deploy patient questionnaires without programming. Automated eDiary scheduling handles daily, weekly, or monthly data capture with one-click invitation management.

Secure, Compliant Patient Registration
Patients register via a secure QR code with no collection of sensitive personal data. A complete audit trail ensures ICH/GCP and 21 CFR Part 11 compliance, fully aligned with GDPR and HIPAA requirements.

Capture With Ease.
Analyze With Confidence.
Collect data in a streamlined, compliant
and efficient way.
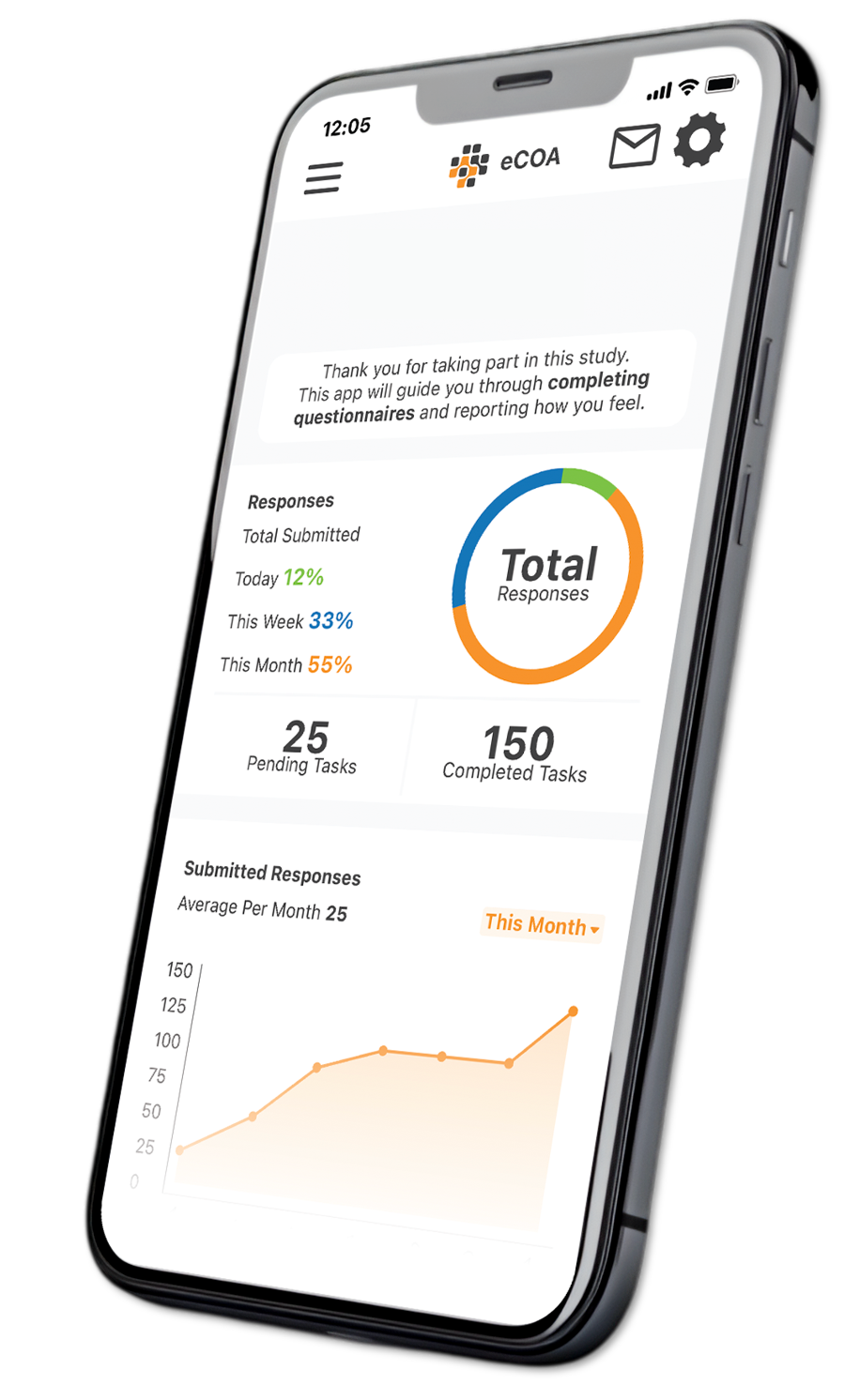
Catchtrial® eCOA is a flexible digital outcome assessment platform supporting intuitive data capture and global instrument deployment, enhancing patient engagement and study execution.

Train and Enable Investigators
Investigators are supported through a customized eCOA app that delivers step-by-step, online guidance for standardized assessments, complemented by on-demand training recordings to ensure consistency across sites.

Standardize Assessments & Scoring
Automated score calculations—such as Detection Threshold (DT)—are performed within the eCOA app, reducing variability, eliminating manual errors, and ensuring consistent data capture across all operators.

Ensure Compliance & Data Readiness
A complete audit trail supports GCP and FDA-compliant data capture, with one-click data exports available through a centralized interface for rapid review, monitoring, and analysis.

Clarity For Patients. Confidence For Research.
Overcome recruitment and consenting challenges by Meditrial proprietary telemedicine-inspired product called Catchtrial® eConsent. eConsent allows researchers to obtain informed consent from participants from anywhere using telemedicine technology.
Catchtrial eConsent enables participants to review and sign informed consent from anywhere using telemedicine-inspired technology. Compatible with Android and iOS, it removes the need for on-site visits and provides a seamless, mobile-first experience for patients.
The app guides participants step by step, presenting study information clearly and assessing comprehension. This patient-centric approach improves engagement, ensures understanding, and enhances data quality across your trial.
Supporting decentralized trial workflows, Catchtrial eConsent significantly reduces the time required for screening and enrollment. Faster, smoother consent processes allow trials to progress efficiently while keeping patients and sites aligned.
Consent from participants or their legally authorized representatives (LARs) is securely captured and stored. The platform maintains a complete audit trail, enabling regulatory compliance and real-time tracking to keep your trial on schedule.

Consent Patients From Anywhere
Catchtrial® eConsent uses telemedicine-inspired technology to obtain informed consent remotely on any Android or iOS device — eliminating the need for on-site visits and accelerating screening and enrollment timelines.

Verify Comprehension Before Enrollment
Built-in quizzed consent guides participants step by step through study information and assesses understanding before signature. Pre-screening surveys evaluate eligibility upfront, reducing screen failures at the site level.

Audit-Ready e-Signatures
Consent from participants or their legally authorized representatives is captured with e-Signatures supported by MFA and biometric verification. A complete audit trail meets 21 CFR Part 11, GDPR, and HIPAA requirements.