In modern clinical trials, the way you capture outcomes is no longer a technical detail—it is a strategic decision that directly affects data quality, regulatory acceptance, and how fast you can move toward approval.
Yet one confusion still creates unnecessary complexity: the difference between eCOA and ePRO.
These terms are often used interchangeably. They should not be.
At Medigen Suite, we see this early in discussions with sponsors. Teams come in thinking they need an “ePRO solution,” only to realize that their study actually requires a broader, coordinated approach across multiple types of outcome data. This is precisely why Medigen Suite was designed from the start as an integrated platform capable of supporting both ePRO and full eCOA within the same environment, without forcing sponsors to stitch together separate tools.
The distinction matters because it shapes everything downstream—from endpoint design to operational execution.
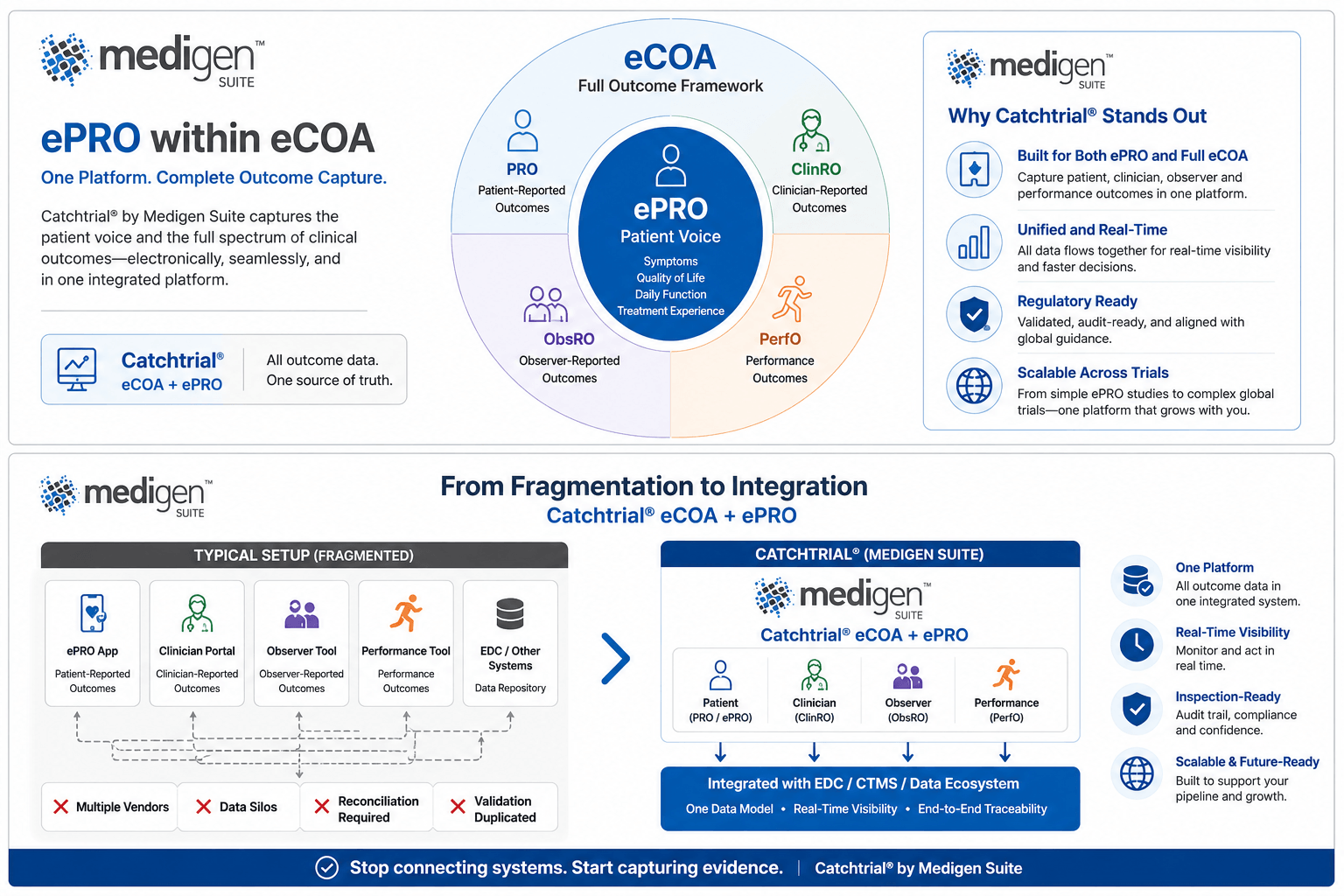
eCOA, or electronic Clinical Outcome Assessment, is the full framework for capturing outcome data digitally. It includes any structured assessment collected electronically in a clinical trial, regardless of who provides the information. This can be the patient, the clinician, a caregiver, or even a standardized performance test. In other words, eCOA represents the complete digital layer of clinical evidence.
ePRO, or electronic Patient-Reported Outcome, is a specific part of that framework. It refers only to data reported directly by the patient, in their own words, without interpretation. When a patient reports pain, fatigue, quality of life, or treatment satisfaction through a mobile app or web interface, that is ePRO. It is one of the most valuable components of modern trials—but it is not the whole picture.
The relationship is simple but important: all ePRO is eCOA, but not all eCOA is ePRO.
This distinction becomes critical when designing a study. If the primary question is centered on how patients feel and function, ePRO may be sufficient and even preferable. However, most pivotal and complex trials require more than the patient’s perspective alone. They require clinical judgment, observable behaviors, and objective performance measures to build a complete and credible assessment of safety and efficacy.
That broader view is what eCOA enables. It brings together four types of outcomes—patient-reported, clinician-reported, observer-reported, and performance-based—into a single, coordinated structure. The challenge is not conceptual; it is operational. Managing multiple data sources, roles, and schedules quickly becomes complex if the underlying technology is fragmented.
This is where many organizations encounter friction. They start with an ePRO tool, then add separate systems for clinician assessments or performance outcomes. Over time, this leads to disconnected workflows, duplicated validation efforts, and inconsistent data structures across studies.
Medigen Suite was designed to avoid exactly this scenario. Instead of treating ePRO as a standalone capability, it embeds it within a broader eCOA-ready ecosystem. This means that whether a study requires only patient-reported data or a full spectrum of outcome assessments, everything is captured, aligned, and monitored within a single platform. The result is not just efficiency—it is consistency, traceability, and inspection readiness across all outcome types.
From a regulatory perspective, this integrated approach is increasingly important. Global guidance emphasizes that endpoints must be fit for purpose and supported by reliable, validated systems. When outcome data flows through multiple disconnected tools, demonstrating that reliability becomes more difficult. A unified platform simplifies validation, strengthens audit trails, and provides a clearer line of sight from data capture to final analysis.
Operationally, the benefits are just as tangible. In a simple ePRO-driven study, Medigen Suite enables intuitive patient engagement, structured schedules, and real-time data visibility without adding unnecessary burden. In more complex trials, the same environment expands to support clinicians, caregivers, and performance-based assessments, all orchestrated according to the protocol. There is no need to change systems as complexity increases, and no need to reconcile data across platforms.
Ultimately, the decision is not whether to choose eCOA or ePRO. Every modern trial uses both, to different degrees. The real question is whether your infrastructure can support that evolution without creating friction.
The most effective sponsors think ahead. They start with a clear understanding of what needs to be measured—not just today, but across their pipeline—and choose a platform that can scale accordingly. This is where the distinction between eCOA and ePRO becomes more than terminology. It becomes a strategic lever.
Clinical trials today are defined by the quality of their evidence and the speed at which that evidence can be trusted. Capturing the patient voice is essential. Capturing the full clinical picture is what makes that evidence actionable.




